Sneha Sudha Komath
Associate Professor,
School of Life Sciences
Jawaharlal Nehru University
New Delhi -110067, India.
Room No. : 406
Off. Phone : 26704502
Mobile: 9868 3320 63
E-mail : sskomath@mail.jnu.ac.in; sskomath@yahoo.com
Education
Ph.D. (1999), University of Hyderabad
2008-present Associate Professor, School of Life Sciences, JNU.
2003-2008 Assistant Professor, School of Life Sciences, JNU.
1999-2002 Lecturer, Department of Chemistry, Panjab University, Chandigarh.
2001-2002 Research Associate, University of Wisconsin, Madison, USA (on leave from PU).
1998-1999 Research Associate, Institute of Microbial Technology, Chandigarh.
1993-1998 JRF/SRF, School of Chemistry, University of Hyderabad.
1991-1993 JRF, SSCU, Indian Institute of Science, Bangalore.
Membership of professional societies
Life member of the Indian Biophysical Society
Member of The American Society of Biochemistry and Molecular Biology (ASBMB)
Awards
National Women Bioscientist’s Award, 2010
Gold Medalist in Chemistry (M.Sc. Univ. of Hyderabad, 1991)
Ranked all-India 4th in AISSE (1984)
Research Supervision
Ph. D. Research: 11 students awarded degree (6 co-supervised); 8 currently working
Project staff: 2
Short-term project trainees: 20+
Current Areas of Research Interest
GPI anchor biosynthesis in human pathogens Candida albicans and Entamoeba histolytica Enzymology and biophysical chemistry
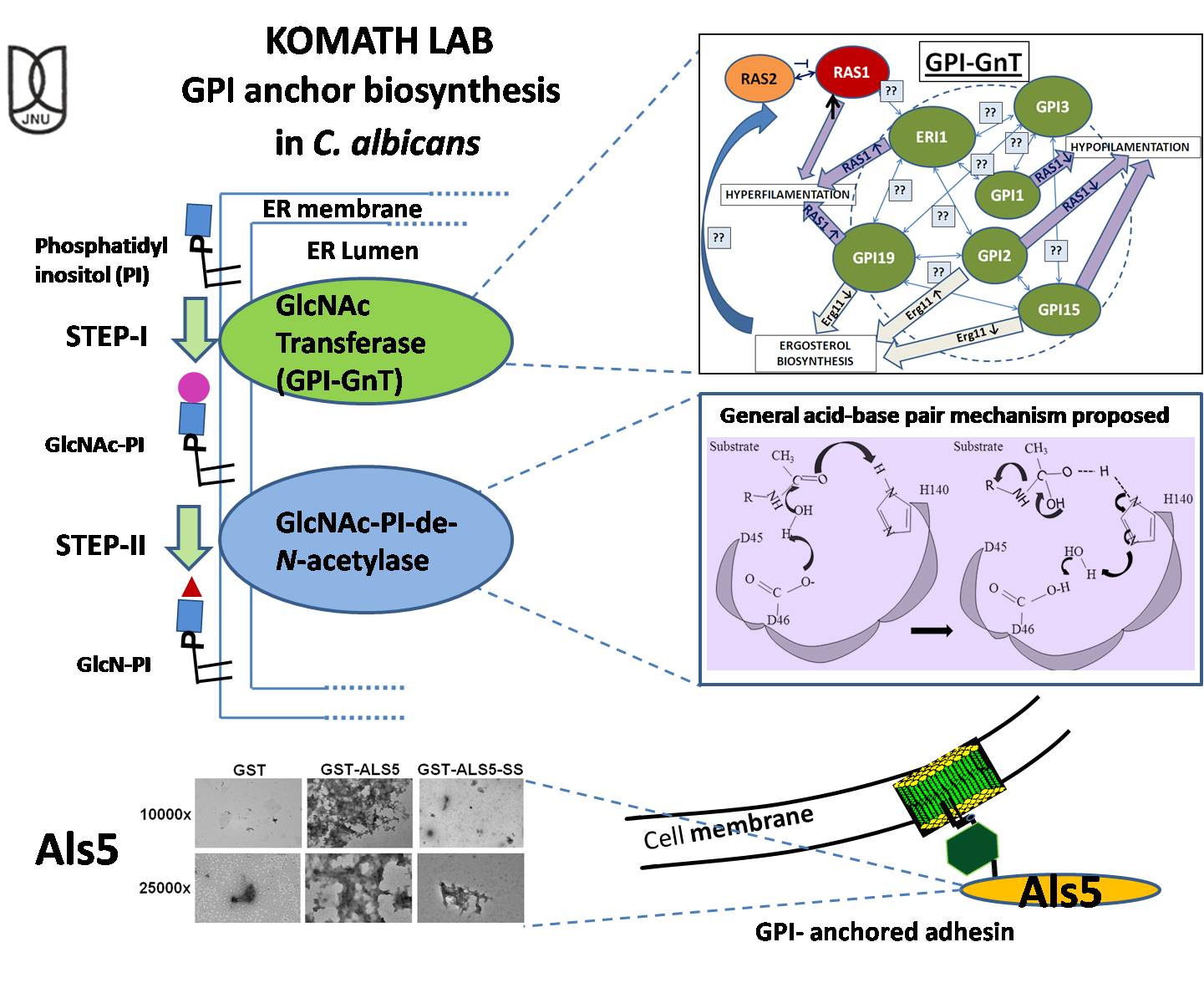
The main focus of the lab currently is glycosylphosphatidylinositol (GPI) anchor biosynthesis in the human pathogen, C. albicans. The GPI anchor is a complex glycolipid anchor that is ubiquitously present in eukaryotes. An elaborate pathway of roughly 10-12 steps works sequentially to produce the precursor GPI anchor in the endoplasmic reticulum. It is then attached to the C-terminal ends of proteins that carry the GPI attachment signal sequence. A variety of proteins may be held by means of this anchor to the extracellular leaflet of the plasma membrane (and/ or cell wall). In lower eukaryotes the biosynthetic pathway leading to the formation of the complete GPI anchor is essential to the viability and functioning of the organism, making it an attractive drug target; in higher eukaryotes it is critical at certain stages of organismal development, such as in embryogenesis, but not at others.
My lab has been working to understand the molecular details of this pathway. This task is particularly challenging given that it involves mostly multi-subunit membrane-bound enzymes. There are no commercially available substrates for most steps of the pathway and assay protocols that are successful for one system do not always work for another system. Listed below are some of our major contributions to the field:
-
We discovered that the enzyme complex, involved in the first step of GPI anchor biosynthesis in C. albicans is mutually co-regulated with ergosterol biosynthesis in the organism and is closely linked to Ras signaling/ hyphal morphogenesis and drug-response in the organism. This has ramifications for controlling Candida infection and countering its drug-resistance. While controlling hyphal morphogenesis is seen as a key step towards controlling virulence in this pathogen, ergosterol and the sterol biosynthetic pathway are the most important targets for therapeutic intervention in controlling Candida infections. Current work in the lab is focused on better understanding the intra-subunit cross-talk as well as the mechanisms of the cross-talk of this step with ergosterol biosynthesis and Ras signaling.
-
We are also interested in comparing the active site of the enzyme involved in the second catalytic step of the pathway, the de-N-acetylation of GlcNAc-PI, between organisms. We showed that the E. histolytica de-N-acetylase is a close homologue of the yeast enzyme and exhibits a unique metal-independent general acid-base pair catalytic mechanism. In contrast the C. albicans homologue shows metal-dependent activity in cell-free systems. Current work is focused on studying the C. albicans and mammalian enzymes.
-
A third line of enquiry in my lab is focused on understanding the role of signal sequences in GPI anchor attachment. We have chosen the C. albicans Als5 protein, an adhesin involved in host recognition, for these studies. We showed that the GPI anchor attachment signal sequence in the Als5 adhesin of C. albicans not only directs GPI anchor attachment but also holds the precursor protein in a conformation that minimizes aggregation and reduces functionality. We have gone on to provide a rationale for this alteration in conformation. This work provides a new paradigm for understanding the importance of signal sequences for GPI anchored proteins. Current work in this project is focused towards better understanding the minimal characteristics of a GPI anchor attachment signal sequence that the transamidase can correctly process for GPI-attachment and to understand whether recognition by the transamidase involves sequence or conformational specificity.
Some new research tools developed:
First in vitro assay for studying GPI biosynthetic activity in C. albicans microsomes. Previously microsomes of C. albicans were always reported to be inactive.
A protocol for expression and purification in high yields of functional de-N-acetylase from the GPI biosynthetic pathway.
Protocols for substrate generation and in vitro assay of the catalytic activity of the de-N-acetylase from amoeba. This method is now working successfully for the mammalian enzyme as well.
Protocols for expression, purification and in vitro analyses of full-length fungal adhesins.
Publications (*Corresponding Author)
- Yadav B, Bhatnagar S, Ahmad MF, Jain P, Pratyusha VA, Kumar P, Komath S.S. * 2014. First step of glycosylphosphatidylinositol (GPI) biosynthesis cross-talks with ergosterol biosynthesis and Ras signaling in Candida albicans. J Biol Chem. 289(6):3365-82. doi: 10.1074/jbc.M113.528802. Epub 2013 Dec 19.
- Ashraf M, Sreejith P., Yadav U. and Komath S. S. *. 2013 Catalysis by N-acetyl-D-glucosaminylphosphatidylinositol de-N-acetylase (PIG-L) from Entamoeba histolytica: New roles for conserved residues. J. Biol. Chem. 288:7590-7595. doi: 10.1074/jbc.M112.427245. Epub 2013 Jan 22.
- Rawal M.K., Khan M.F., Kapoor K., Goyal N., Sen S., Saxena A.K., Lynn A.M., Tyndall J.D.A., Monk B.C., Cannon R.C., Komath S. S., and Prasad R. 2013. Insight into PDR ABC pump drug transport through mutagenesis of Cdr1p transmembrane domains. J. Biol. Chem. 288(34):24480-93. doi: 10.1074/jbc.M113.488353. Epub 2013 Jul 3.
- Victoria G.S., Yadav B., Hauhnar L., Jain P., Bhatnagar S.and Komath S.S.*. 2012. Mutual Co-Regulation Between GPI-N-acetylglucosaminyl Transferase and Ergosterol Biosynthesis in Candida albicans. Biochem. J. May 1;443(3):619-25. (Identified by the Target Selection Group of Global Medical Discovery as a paper of particular interest for drug discovery: http://globalmedicaldiscovery.com/key-scientific-articles)
- Ahmad M. F., Yadav B., Kumar P., Puri A., Mazumder M., Ali A., Samudrala S., Muthuswami R., and Komath S. S. *. 2012. The GPI anchor signal sequence dictates the folding and functionality of the ALS5 adhesin from Candida albicans. PLoS One 7(4):e35305. (Identified by the Target Selection Group of Global Medical Discovery as a paper of particular interest for drug discovery: http://globalmedicaldiscovery.com/key-scientific-articles).
- Dutta P., Tanti G.K., Sharma S., Goswami S.K., Komath S.S., Mayo M.W., Hockensmith J.W., Muthuswami R. 2012. Global Epigenetic Changes Induced by SWI2/SNF2 Inhibitors Characterize Neomycin-Resistant Mammalian Cells. PLoS One 7(11):e49822.
- Nongkhlaw M., Gupta M., Komath S. S. and Muthuswami R. 2012. Motifs Q and I are required for ATP hydrolysis but not for ATP binding in SWI2/SNF2 proteins. Biochemistry 51(18):3711-22.
- Ashraf M., Yadav B., Sreejith, Kumar K. S., Vats D., Muthuswami R. and Komath S. S.*. 2011. The N-Acetyl-D-glucosaminylphosphatidylinositol de-N-acetylase from Entamoeba histolytica: Metal alters catalytic rates but not substrate affinity. J. Biol. Chem. 286: 2543-2549
- Singh P, Verma P, Yadav B, Komath S.S.* 2011. Synthesis and evaluation of indole-based new scaffolds for antimicrobial activities-Identification of promising candidates. Bioorg Med Chem Lett. 2011 Jun 1;21(11):3367-72.
- G. S. Victoria, P. Kumar and Komath S. S. *. 2010. The Candida albicans homolog of PIG-P: Gene dosage and role in growth and filamentation. Microbiology. 156, 3041-3051.
- Singh, P., Kaur,J. Yadav, B. and Komath, S. S.* 2010. Targeting efflux pumps—In vitro investigations with acridone derivatives and identification of a lead molecule for MDR modulation. Bioorg. Med. Chem. 18 (2010) 4212–4223.
- Pandey, G., Fatma, T., Cowsik, S. M. and Komath S. S.* 2009. Specific interaction of jacalin with phycocyanin, a fluorescent phycobiliprotein. J. Photochem. Photobiol. B. Biol. 97(2):87-93.
- Pandey, G., Fatma, T and Komath S. S.* 2009. Specific interaction of the legume lectins, concanavalin A and peanut agglutinin, with phycocyanin. Photochem. Photobiol. 85(5):1126-33.
- Singh, P., Kaur,J. Yadav, B. and Komath, S. S.* 2009. Design, synthesis and evaluation of acridone derivatives using Candida albicans- Search for MDR modulators led to identification of an anti-candidiasis agent. Bioorg. Med. Chem. 17(11):3973-9. Epub 2009 Apr 18.
- Nongkhlaw, M., Jha, D. K., Hockensmith, J.W., Komath, S.S.*and Muthuswami, R.* 2009. Elucidating the mechanism of DNA-dependent ATP hydrolysis mediated by DNA-dependent ATPase A, a member of the SWI2/SNF2 protein family. Nucleic Acid Research 37(10):3332-41. Epub 2009 Mar 26.
- Oswal N, Sahni NS, Bhattacharya A, Komath S.S.*, Muthuswami R.* 2008. Unique motifs identify PIG-A proteins from glycosyltransferases of the GT4 family. BMC Evol Biol. Jun 4;8:168.
- Rai V, Gaur M, Kumar A, Shukla S, Komath S.S.*, Prasad R.* 2008. A novel catalytic mechanism for ATP hydrolysis employed by the N-terminal nucleotide-binding domain of Cdr1p, a multidrug ABC transporter of Candida albicans. Biochim Biophys Acta. (Biomembranes) 1778(10):2143-53.
- Rai, V., Gaur M., Shukla S., Shukla, S., Ambudkar S.V., Komath S. S. and Prasad, R. 2006. Conserved Asp327 of Walker B motif in the N-terminal Nucleotide Binding Domain (NBD-1) of Cdr1p of Candida albicans has acquired a new role in ATP hydrolysis. Biochemistry. 45(49):14726-39.
- Komath S. S.*, Kavitha M., Swamy M. J.* 2006. Beyond carbohydrate binding: New directions in plant lectin research. Org Biomol Chem. 4(6):973-88. Most downloaded paper of the month.
- R. Prasad, N. A. Gaur, M. Gaur and S. S. Komath. 2005. Efflux Pumps in Drug Resistance of Candida. Infect Disord Drug Targets. 6(2):69-83.
- Saini P., Prasad T., Gaur N. A., Shukla S., Jha S., Komath S. S., Khan L. A., Haq Q. M. R. and Rajendra Prasad. 2005. Alanine scanning of transmembrane helix 11 of Cdr1p ABC antifungal efflux pump of Candida albicans: identification of amino acid residues critical for drug efflux. J Antimicrob Chemother. 56:77-86.
- Rai, V., Shukla, S., Jha, S., Komath S. S.* and Prasad, R.* 2005 Functional characterization of N-terminal nucleotide binding domain (NBD-1) of a major ABC drug transporter Cdr1p of Candida albicans: Uncommon but conserved Trp326 of Walker B is important for ATP binding. Biochemistry. 44: 6650-6661.
- Kenoth, R., Komath S. S. and Swamy, M. J. 2003. Physicochemical and saccharide-binding studies on the galactose-specific seed lectin from Trichosanthes cucumerina. Arch. Biochem. Biophys. 413(1):131-138.
- Manoj , N,. Jeyaprakash, A. A., Pratap, J. V., Komath, S. S., Kenoth, R., Swamy, M. J., Vijayan, M. 2001. Crystallization and preliminary X-ray studies of snake gourd lectin: homology with type II ribosome-inactivating proteins. Acta Crystallogr. D Biol. Crystallogr. 57:912-9144.
- Komath, S. S., Kenoth , R,. Swamy, M. J. 2001. Thermodynamic analysis of saccharide binding to snake gourd (Trichosanthes anguina) seed lectin. Fluorescence and absorption spectroscopic studies. Eur J Biochem. 268:111-119.
- Komath, S. S., Bhanu, K., Maiya, B. G. and Swamy, M. J. 2000. Binding of porphyrins to jacalin [jack fruit (Artocarpus integrifolia) agglutinin]. Absorption and fluorescence spectroscopic investigations. Biosci. Rep. 20:265-276.
- Komath, S. S., R. Kenoth, Giribabu, L., Maiya, B. G. and Swamy, M. J. 2000. Fluorescence and absorption spectroscopic studies on the interaction of porphyrins with snake gourd (Trichosanthes anguina) seed lectin. J. Photochem. Photobiol. (B. Biology) 55:49-55.
- Chaudhary, A., Vasudha, S., Rajagopal, K., Komath, S. S., Garg, N., Yadav, M., Mande, S. C. and Sahni, G. 1999. Function of the central domain of streptokinase in substrate plasminogen docking and processing as revealed by site-directed mutagenesis. Protein Science, 8:2791-2805.
- Padma, P., Komath, S. S., Nadimpalli, S. K. and Swamy, M. J. 1999. Purification in high yield and characterisation of a new galactose-specific lectin from the seeds of Trichosanthes cucumerina. Phytochemistry, 50: 363-371.
- Komath, S. S. and Swamy, M. J. 1998. Fluorescence quenching, time resolved fluorescence and chemical modification studies on the tryptophan residues of snake gourd (Trichosanthes anguina) seed lectin. J. Photochem. Photobiol.(B. Biology) 50:108-118.
- Padma, P., Komath, S. S., Swamy, M. J. 1998. Fluorescence quenching and time-resolved fluorescence studies on the Momordica charantia (bitter gourd) seed lectin. Biochem. Mol. Biol. Int. 45(5), 911-920.
- Komath, S. S., Nadimpalli, S. K. and Swamy, M. J. 1998. Identification of histidine residues in the sugar binding site of snake gourd (Trichosanthes anguina) seed lectin. Biochem. Mol. Biol. Int. 39(2):243-252.
- Komath S. S. and Swamy, M. J. 1998. Further characterisation of the saccharide specificity of snake gourd (Trichosanthes anguina) seed lectin. Current Science, 75(6): 608-611.
- Bhanu, K., Komath, S. S. and Swamy, M. J. 1997. Interaction of porphyrins with concanavalin A and pea lectin. Current Science. 73(7):598-602.
- Ramakrishnan, M. Komath, S. S., Sheeba, V. and Swamy, M. J. 1997. Differential scanning calorimetric studies on the thermotropic phase transitions of dry and hydrated forms of N-acylethanolamines of even chainlengths. Biochim. Biophys. Acta. 39(2):243-252.
- Komath, S. S., Nadimpalli, S. K. and Swamy, M. J. 1996. Purification in high yield and characterisation of the galactose-specific lectin from the seeds of snake gourd (Trichosanthes anguina). Biochem. Mol. Biol. Int. 39(2):243-252.
- Vijayadamodar, G. V., Komath, S. S., Roy, S. and Bagchi, B. 1994. Dielectric relaxation in dipolar solid rotator phases. Phase Transitions. 50:21-45.
- Roy, S., Komath, S. S. and Bagchi, B. 1993. Molecular theory of ultrafast solvation in liquid acetonitrile. J. Chem. Phys. 99(4):3139-3058.
- Komath, S. S. and Bagchi, B. 1993. Solvation dynamics in a Brownian dipolar lattice. Comparison between computer simulation and various molecular theories of solvation dynamics. J. Chem. Phys. 98(11):8987-8993.
- Roy, S., Komath, S. S. and Bagchi, B. 1993. Dielectric friction and solvation dynamics: novel results on relaxation in dipolar liquids. Proc. Indian Acad. Sci. (Chem. Sci.). 105(1): 79-85.
- Chattopadhyay, A., Komath, S. S. and Raman, B. 1992. Aggregation of lasalocid A in membranes: a fluorescence study. Biochim. Biophys. Acta. 1104:147-150.
Book Chapter (peer-reviewed)
- Komath S. S.*, Ahmad F. A. and Mazumder M. 2013. Cloning, expression and functional characterization of Als5, an adhesin from Candida albicans. Protein Purification and Analysis – Methods and Applications. ISBN: 978-1477555-05-7. iConcept Press.
Popular Science Commentary:
- Komath S. S.* 2008. Frames of science? Current Science 94(11): 1363-64.
Funded Research Projects
Completed: 6 as Principal Investigator (PI)
Current Projects (as PI):
GPI biosynthesis and ras signaling in Candida albicans. Funded by Department of Biotechnology, India.
Cross-talk between GPI biosynthesis and Ras signaling in S. cerevisiae. Sanctioned by DST (yet to begin).
Mammalian PIG-L: Cloning, purification and characterization of GlcNAc-PI de-N-acetylase activity. Sanctioned by CSIR (yet to begin).
Some Recent Invited Talks:
-
On the trail of a novel de-N-acetylase on 9th April, 2013 at Department of Biotechnology, Lund University Sweden.
-
Some like it fat and sweet. Glycolipid anchors on proteins at TRendy’s in Biochemistry, 2013 (held this year at University of Hyderabad, Hyderabad).
-
Multitasking the fungal way: GPI biosynthesis, ergosterol biosynthesis and hyphal growth in Candida albicans at 20 Years of CDR1 Research, 4th-8th Jan, 2014, JNU, New Delhi.
-
A novel de-N-acetylase from E. histolytica that uses a metal-independent catalytic mechanism to generate glucosaminyl phosphatidylinositol from N-acetylglucosaminyl phosphatidylinositol (GlcNAc-PI) at International Carbohydrate Symposium satellite symposium on Emerging Trends in Glycoscience and Glycotechnology (ETGG-2014), IIT Delhi, 8th-10th Jan 2014.
-
Novel cross-talk of glycosylphosphatidylinositol (GPI) anchor biosynthesis with ergosterol and Ras signalling in Candida albicans at 27th International Carbohydrate Symposium, 12-17th Jan 2014, Bangalore
-
A signal with a difference: The role of GPI anchor signal sequence in dictating conformation and function of the Als5 adhesin in Candida albicans at IUBMB 10th International Symposium on Cell Surface Macromolecules, Jan20-24th 2014, Kolkata.
-
Ergosterol sensing and Ras signalling in Candida albicans. Vistas of Life Sciences, Now and Beyond (Celebrating four decades of School of Life Sciences, JNU), 13-15th Feb 2014.